|
Total number of valence electrons for SF2 – Valence electrons of Sulphur + Valence electrons of Fluorine So we will first find out the total valence electrons for Sulphur Difluoride. You are watching: Does sf2 have a dipole momentįor drawing the Lewis structure for any molecule, we first need to know the total number of valence electrons. 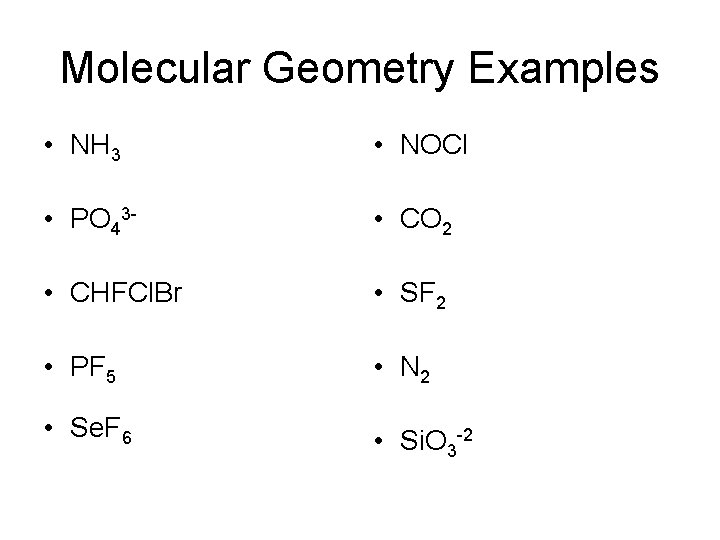
In this blog post, we will look at the Lewis dot structure of SF2, its molecular geometry and shape. It has a chemical formula of SF2 and can be generated by the reaction of Sulphur Dioxide and Potassium Fluoride or Mercury Fluoride. 
Sulfur Difluoride is an inorganic molecule made up of one Sulphur atom and two Fluorine atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
